 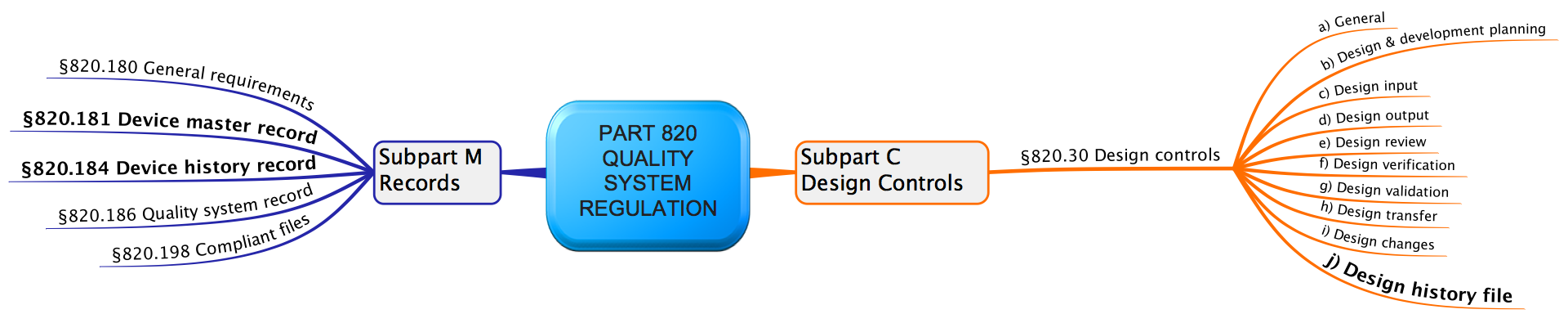
The Device Master Record (DMR) can be considered an instruction manual for the compliant manufacture of the medical device. The 21 CFR 820.30, states that “each manufacturer shall establish and maintain a DHF for each type of device.” 
A DHF must contain or refer to the documentation of design reviews, validation, controlled design, and approved design inputs and output. The Design History File (DHF) documents how the instruction manual was designed and compiled. What Do DHF, DHR, and DMR Stand For? DHF - Design History File They are the required documents that medical device manufacturers should create and make available for auditors to show the compliance of the medical device to achieve FDA approval - and therefore access to the US market. These three abbreviations are commonly used by the FDA in medical device quality regulation:Īlthough they sound almost the same, they are quite different and can contain different documents generated in different steps of the design process. And as a medical device professional you can easily get lost in this acronym ocean! The most important (and most confusing) abbreviations are DHF, DMR, and DHR. 
In the medical device industry, there are tons of abbreviations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
